While peptides incorporating a single metal-binding site are now designed with relative ease, coordination of multiple metal ions is more challenging. 18 Additionally, the Peacock lab has generated a trimeric coiled-coil peptide that employs asparagine and aspartic acid residues to bind lanthanides. Other notable examples of coiled-coil-based peptides which bind a single metal ion include a homodimeric Cd( ii)-binding mimic of the protein rubredoxin from the Ogawa lab 16 a Zn( ii)-binding heterodimer from the Jerala lab 17 and a Zn( ii) binding tetrameric helical bundle from the Kuhlman and Hilvert labs. 13 The Tanaka lab have also made significant contributions to this field with their trimeric IZ peptide, which contains two histidine residues and binds Co( ii), Cu( ii), Ni( ii) and Zn( ii), 14 and their tetrameric AM2D and AM2E peptides which also bind Cu( ii) and Zn( ii).
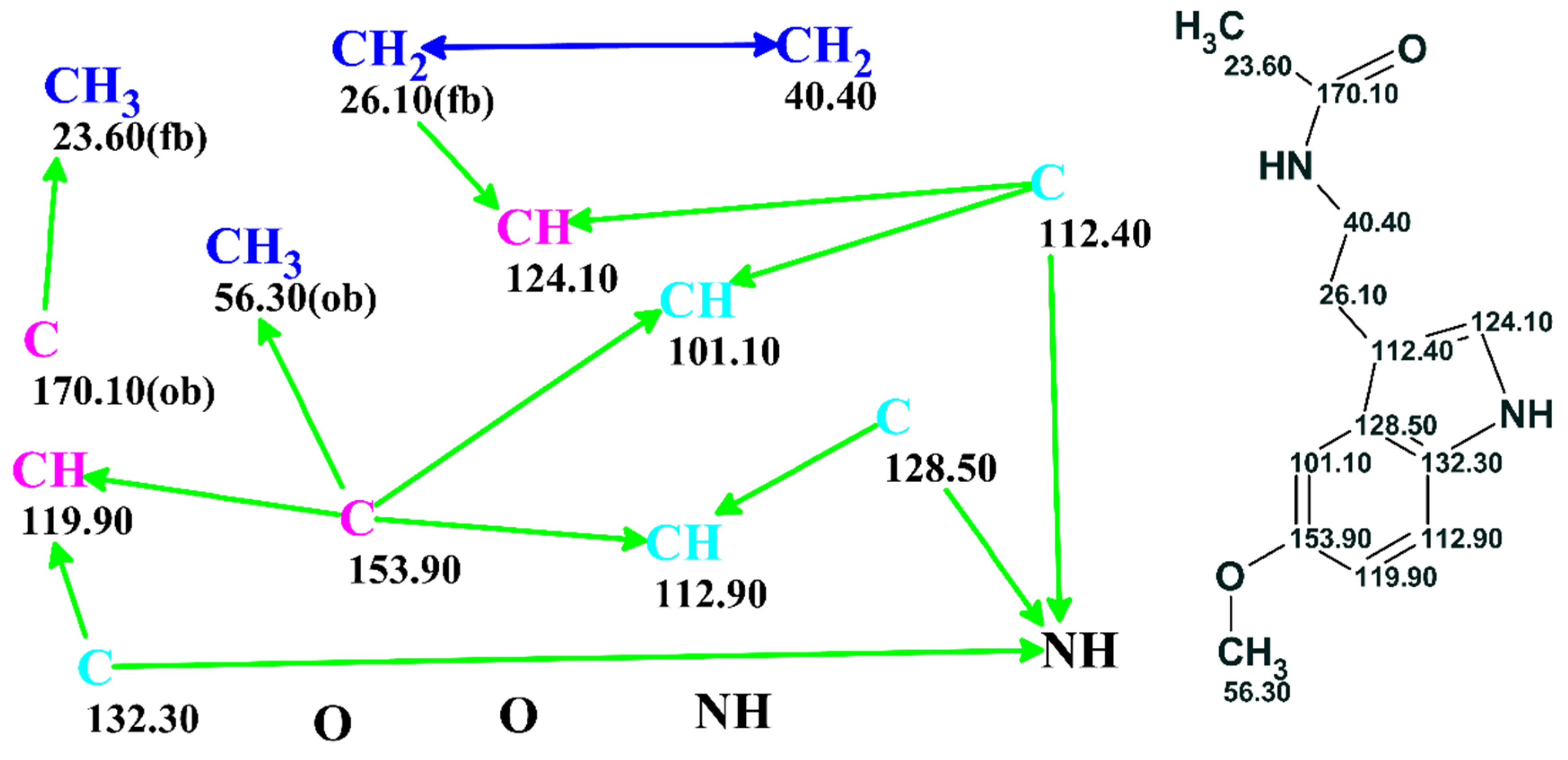
AUTO ASSIGN CARBONS MESTRENOVA SERIES
The Pecoraro lab in particular have presented multiple designs of this type, utilising their TRI series of peptides, 6– 8 so called as they form trimeric coiled coils, and variants of α3D 9– 12 a single-chain three-helix bundle originally developed by the DeGrado lab. The most common examples of metal-binding coiled coils substitute hydrophobic residues at the ‘ a’ and ‘ d’ positions with amino acids capable of coordinating metal ions, and this often results in the binding of a single metal ion in the hydrophobic core of the coiled coil. 2, 3 These characteristics have enabled researchers to devise rules that allow coiled coils with prescribed oligomer states and orientations to be reliably designed, 4, 5 making them attractive candidates for metallopeptide design.

1 Most coiled coils also have a repeating sequence pattern known as a ‘heptad repeat,’ which is given the nomenclature abcdefg, with hydrophobic residues found at the ‘ a’ and ‘ d’ positions and polar residues occupying the other positions. Canonical coiled coils are formed when two or more α-helices interact in a specific manner, known as ‘knobs-into-holes’ packing. One scaffold that has been widely employed for this purpose is the α-helical coiled coil. Additionally, HisAD demonstrates an unprecedented discrimination between transition metal ions, the basis of which is likely to be related to the stability of the peptide-metal complexes formed.Ī fundamental aim of metalloprotein design is to understand how metal-binding and protein folding affect each other and to use this knowledge to produce novel metallopeptides, often utilising minimal scaffolds. The X-ray crystal structure of the HisAD-Cu( ii) complex reveals the trimeric HisAD peptide coordinates three Cu( ii) ions this is the first example of such a structure. These show the peptide is trimeric and binds to both Cu( ii) and Ni( ii) in a 1 : 1 ratio with the histidine residues involved in the metal coordination, as designed. The structure, and metal-binding ability, of HisAD is probed using a combination of circular dichroism (CD) spectroscopy, analytical ultracentrifugation (AUC), nuclear magnetic resonance (NMR) spectroscopy and X-ray crystallography. HisAD, the resulting peptide, is unstructured in the absence of metal and folds selectively to form an α-helical construct upon complexation with Cu( ii) and Ni( ii) but not Co( ii) or Zn( ii). Here, histidine residues are introduced into the hydrophobic core of a coiled-coil trimer, generating a peptide that self-assembles upon the addition of metal ions. Designing peptides that fold and assemble in response to metal ions tests our understanding of how peptide folding and metal binding influence one another.
